Transmembrane proteins are among the most valuable yet technically challenging drug targets, accounting for a significant proportion of therapeutically relevant receptors, transporters, and signaling molecules. However, their complex structures, multiple hydrophobic transmembrane domains, and strict conformational requirements make them extremely difficult to express, purify, and present in a native, functional form.
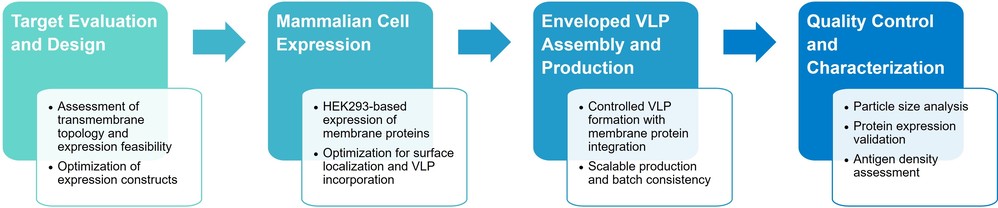
Profacgen's Enveloped Virus-Like Particle (VLP) Technology provides an advanced solution for membrane protein production by displaying correctly folded, full-length transmembrane proteins in their native lipid bilayer environment. Built on a robust HEK293 expression platform, our enveloped VLPs enable high-density, conformationally intact antigen presentation, significantly improving antibody discovery success rates. This platform is ideally suited for immunization, antibody screening, and biophysical analysis of complex membrane proteins that are otherwise inaccessible using conventional expression methods.
Transmembrane proteins are embedded in the phospholipid bilayer of cellular membranes, spanning intracellular and extracellular spaces to mediate signal transduction, molecular transport, immune recognition, and cellular communication. Their interactions with extracellular ligands and intracellular signaling partners are central to physiological regulation, and dysfunctions in these proteins are closely associated with cancer, autoimmune diseases, neurological disorders, and infectious diseases.
Despite their importance, recombinant production of transmembrane proteins remains a major bottleneck. Many multi-pass membrane proteins contain multiple hydrophobic regions that severely limit expression levels in host cells and often result in misfolding, aggregation, or loss of biological activity. Traditional solubilization and purification strategies—regardless of antibody capture or affinity methods—frequently disrupt native conformations, leading to poor antigen quality and low success rates in antibody screening.
For antibody discovery, especially therapeutic antibody development, it is essential that antigens retain native conformation, proper orientation, and full biological activity. The inability to reliably prepare such antigens has become an urgent and widely recognized challenge.
Virus-like particles (VLPs) are self-assembled nanostructures composed of viral structural proteins but lacking viral genetic material, making them non-infectious and inherently safe. Enveloped VLPs further incorporate a lipid bilayer derived from host cell membranes, closely mimicking the natural environment of membrane proteins.
In Profacgen's Enveloped VLPs platform, target transmembrane proteins are expressed in mammalian cells and naturally incorporated into the VLP membrane during particle assembly. As a result, membrane proteins are presented in their full-length form, correct topology, and native lipid context, without the need for harsh detergents or artificial refolding.
 Figure 1. Overview of enveloped VLPs technology for membrane protein production. (Adapted from Nooraei et al., 2021)
Figure 1. Overview of enveloped VLPs technology for membrane protein production. (Adapted from Nooraei et al., 2021)
This approach converts difficult-to-handle membrane proteins into soluble, high-concentration, nanoparticle-displayed antigens, ideally suited for antibody immunization and screening workflows.
Profacgen has established a specialized Enveloped VLPs technology platform based on the HEK293 mammalian expression system, ensuring high fidelity protein folding and post-translational modification.
Background
A biotechnology client needed functional antibodies against a highly conserved GPCR with seven transmembrane domains. This target was notoriously difficult to handle; conventional detergent-solubilized antigens consistently collapsed native epitopes, yielding antibodies with poor specificity and no receptor-modulating activity.
Our Solution
We produced enveloped virus-like particles displaying the full-length GPCR in its native phospholipid bilayer. This approach preserved the receptor's conformational integrity and lateral mobility while achieving high antigen density per particle. The VLPs were stable, reproducible, and administered directly for mouse immunization without additional formulation.
Final Results
The campaign successfully generated multiple antibody lineages recognizing the receptor in its native state. Several candidates demonstrated potent ligand-blocking activity in cell-based assays. By delivering authentic membrane antigen architecture rather than destabilized protein fragments, we enabled the client to discover therapeutically relevant antibodies that had been inaccessible using traditional recombinant approaches, dramatically accelerating their biologics pipeline.
Background
A pharmaceutical company required a native ion channel antigen to support high-throughput antibody screening and binding kinetics profiling. Previous efforts using purified, detergent-extracted protein failed repeatedly: the multi-pass channel aggregated upon solubilization, and binding signals were irreproducible.
Our Solution
We applied our enveloped VLP platform to incorporate the full-length ion channel into intact membrane bilayers. The channel inserted with correct topology and retained activity without requiring detergents or reconstitution steps. VLPs were directly compatible with both ELISA and biolayer interferometry platforms, producing strong, dose-responsive binding curves.
Final Results
Multiple high-affinity antibodies were successfully identified, each confirmed to recognize the channel's native conformation. These hits enabled downstream functional assays and target validation studies that had previously been unattainable. By preserving the antigen in a near-native lipid environment, we eliminated the performance gap between recombinant production and biological relevance, allowing the client to advance confidently into lead optimization.
Background
An academic consortium investigating viral entry mechanisms required an envelope protein presented with authentic membrane anchoring and native trimeric architecture. Epitope integrity was strictly dependent on proper lipid bilayer integration, yet live virus handling posed safety and containment challenges.
Our Solution
We generated enveloped VLPs displaying the full-length viral transmembrane protein in its native orientation and oligomeric state. The particles mimicked infectious virion surface topology while completely eliminating genetic material, providing a biosafe reagent suitable for standard BSL-2 facilities.
Final Results
Immunization studies demonstrated robust, antigen-specific humoral responses, and the VLPs enabled efficient functional screening of neutralizing antibodies. The platform preserved quaternary epitopes lost in soluble ectodomain preparations and supported both vaccine efficacy studies and discovery of entry-blocking biologics. By combining authentic antigen presentation with an enhanced safety profile, we provided the consortium a versatile tool spanning fundamental virology through translational antibody development.
Consult Our Experts on Your Project
Committed to advancing global antibody drug innovation, Profacgen provides comprehensive enveloped VLPs services as part of its advanced recombinant protein and antibody R&D platform. Contact us today to discuss your project with our experts!
Reference:
Fill out this form and one of our experts will respond to you within one business day.